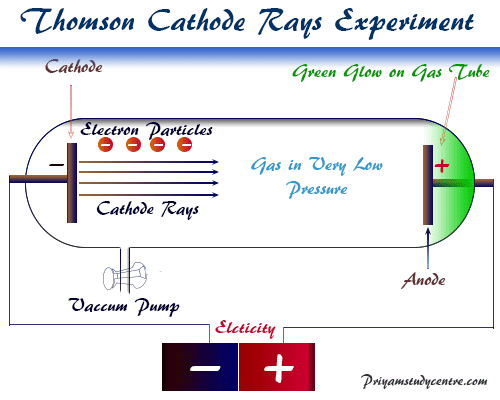
So, the charge of the electron will be -1.602 x 10-19 C. This value is then used to calculate a new approximation to A r(e), and the process repeated until the values no longer vary (given the relative uncertainty of the measurement, 2.1×10 −9): this happens by the fourth cycle of iterations for these results, giving A r(e) = 5.485799111(12)×10 −4 for these data. It is a fundamental physical constant that is used to express the naturally occurring unit of electric charge, which is 1.602 × 10-19 coulomb. This approximate value is then used to calculate a first approximation to A r( 12C 6+), knowing that E b( 12C)/ m u c 2 (from the sum of the six ionization energies of carbon) is 1.1058674×10 −6: A r( 12C 6+) ≈ 11.9967087236367. Such correction is only substantial for electrons accelerated by voltages of well over 100 kV.įor example, the relativistic expression for the total energy, E, of an electron moving at speed \displaystyleĪs the relative atomic mass of 12C 6+ ions is very nearly 12, the ratio of frequencies can be used to calculate a first approximation to A r(e), 5.4863037178×10 −4. Thomsons experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. The fundamental unit of charge is often represented as e. If the electron is moving at a relativistic velocity, any measurement must use the correct expression for mass. The same number of electrons is required to make 1.00 C of electric charge. 
The charge of an electron is 1.6×1019 and the charge of proton is +1.6×10. Protons are positively charged and are present in the nucleus. 
Electrons are negatively charged and revolve around the nucleus. Most practical measurements are carried out on moving electrons. Electrons and protons are the subatomic particles of an atom. 
Beyond, it opens the way to further developments in metrology and in fundamental physics, such as a quantum multimeter or new accurate comparisons to single electron pumps.The term "rest mass" is sometimes used because in special relativity the mass of an object can be said to increase in a frame of reference that is moving relative to that object (or if the object is moving in a given frame of reference). This new quantum current source, able to deliver such accurate currents down to the microampere range, can greatly improve the current measurement traceability, as demonstrated with the calibrations of digital ammeters. A possibility is given by a Zitterbewegung interpretation of quantum mechanics, according to which charged elementary particles can be modeled by a current ring. elementary charge expressed in coulombs). On the submicroscopic scale, it is more convenient to define an energy unit called the electron volt (eV), which is the energy given to a fundamental charge accelerated through a potential difference of 1 V. Brun-Picard and 3 other authors Download PDF Abstract:One major change of the future revision of the International System of Units (SI) is a new definition of the ampere based on the elementary charge \emph$. 1 eV 1.602176487(40)×1019 J (the conversion factor is numerically equal to the. Download a PDF of the paper titled Practical quantum realization of the ampere from the electron charge, by J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
